|
The lower figure of 2.8 months increased survival in interim phase III results is a probable result of stopping the trial median survival time for patients still alive could not be calculated. Earlier phase II of the trial showed a median increased survival of 18.9 weeks (around 4.4 months). The ALSYMPCA study was stopped early after a pre-planned efficacy interim analysis, following a recommendation from an Independent Data Monitoring Committee, on the basis of achieving a statistically significant improvement in overall survival (two-sided p-value = 0.0022, HR = 0.699, the median overall survival was 14.0 months for 223Ra and 11.2 months for placebo). Ģ23Ra successfully met the primary endpoint of overall survival in the phase III ALSYMPCA (ALpharadin in SYMptomatic Prostate CAncer patients) study for bone metastases resulting from CRPC in 922 patients. The phase II study of radium-223 in castration-resistant prostate cancer (CRPC) patients with bone metastases showed minimum myelotoxicity and good tolerance for the treatment. Clinical trials and FDA and EMA approval Taking account of its preferential uptake by bone and the alpha particles' short range, radium-223 is estimated to give targeted osteogenic cells a radiation dose at least eight times higher than other non-targeted tissues. This reduces damage to surrounding healthy tissues, producing an even more localized effect than the beta-emitter strontium-89, also used to treat bone cancer. Alpha radiation has a very short range in tissues compared to beta or gamma radiation: around 2–10 cells. Although radium-223 and its decay products also emit beta and gamma radiation, over 95% of the decay energy is in the form of alpha radiation. Radium is preferentially absorbed by bone by virtue of its chemical similarity to calcium, with most radium-223 that is not taken up by the bone being cleared, primarily via the gut, and excreted. The use of radium-223 to treat metastatic bone cancer relies on the ability of alpha radiation from radium-223 and its short-lived decay products to kill cancer cells. See also: Targeted alpha-particle therapy Medical use Radium-223 chloride Clinical data Ģ23Ra itself decays to 219Rn (half-life 3.96 s), a short-lived gaseous radon isotope, by emitting an alpha particle of 5.979 MeV. This decay path makes it convenient to prepare radium-223 by "milking" it from an actinium-227 containing generator or "cow", similar to the moly cows widely used to prepare the medically important isotope technetium-99m. Actinium-227 (half-life 21.8 years) in turn decays via thorium-227 (half-life 18.7 days) to radium-223. 
Origin and preparation Īlthough radium-223 is naturally formed in trace amounts by the decay of uranium-235, it is generally made artificially, by exposing natural radium-226 to neutrons to produce radium-227, which decays with a 42-minute half-life to actinium-227. The principal use of radium-223, as a radiopharmaceutical to treat metastatic cancers in bone, takes advantage of its chemical similarity to calcium, and the short range of the alpha radiation it emits. Radium-223 dichloride is an alpha particle-emitting radiotherapy drug that mimics calcium and forms complexes with hydroxyapatite at areas of increased bone turnover. 
Godlewski, a Polish chemist from Kraków, and was historically known as actinium X (AcX). Patients will be treated with radium-223 according to standard of care, while carefully being monitored by obtaining multi-parametric parameters from blood, imaging and tissue.Radium-223 ( 223Ra, Ra-223) is an isotope of radium with an 11.4-day half-life. Within the Radium223Insight, 30 patients will be included. The Radium223Insight is a collaborative effort of the Erasmus MC Rotterdam, Radboud UMC Nijmegen and the Franciscus Gasthuis & Vlietland Hospital, Rotterdam. 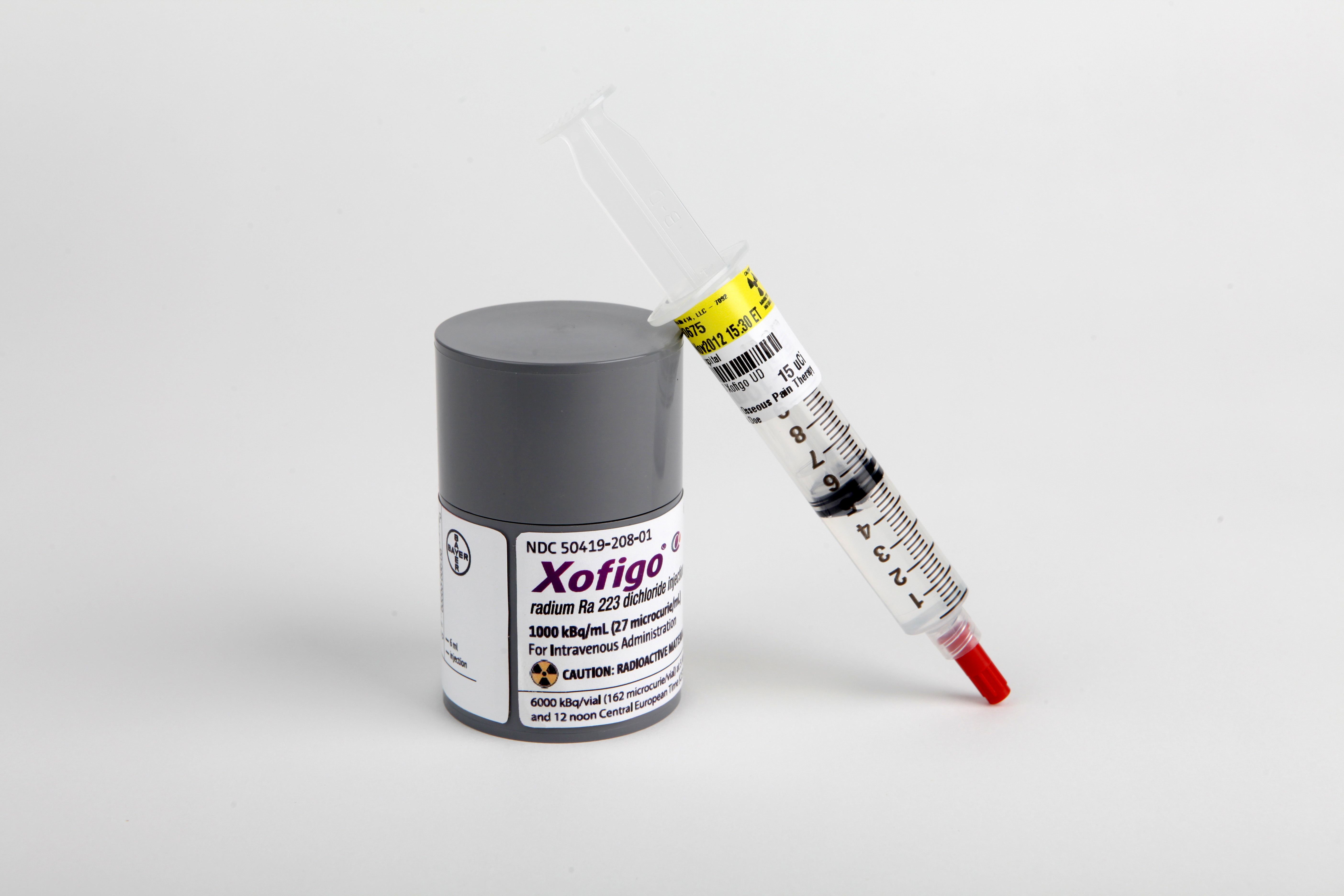
The Radium223Insight study aims to identify biomarkers for early identification of clinical benefit from radium-223 treatment and to better understand immune response during radium-223 treatment in mCRPC patients. In addition, previous studies have shown that radium-223 can induce immune responses, indicating that radium-223 may improve the efficacy of immune checkpoint inhibition. However, as traditional parameters, including PSA, fail in (early) response evaluation, other parameters are needed to guide treatment planning in mCRPC patients, treated with radium-223. Based on the survival results of a randomized phase 3 trial in 2013, the European Medicines Agency (EMA) has approved radium-223 for the treatment of metastatic castration resistant prostate cancer (mCRPC) with bone metastases only.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
